Abstract
Iron overload is a potentially preventable complication of blood transfusion dependency. The effectiveness and safety of early start of iron chelation therapy in young children with transfusion-dependent Beta-thalassemia (TDB-T) prior to development of iron overload have been recently demonstrated. The aim of this study was to evaluate the long term effectiveness and safety of continued iron chelation in these children. Methodology: This is a multi-center, investigator initiated, one year prospective randomized study of children with TDB-T who completed the randomized START study (NCT03591575), which had evaluated the safety and efficacy of the oral iron chelator (DFP) in children who did not yet meet the criteria for starting chelation therapy as per standard practice. Enrolment is shown in follow-up chart below; 48 children with TDBT from 3 centers were eligible to be enrolled and were randomized in 1:1 ratio to receive either DFP (n=23) or deferasirox (DFX) (n=25). Half of patients in this study were naïve to chelation (on placebo in START study); had received DFP at a dose; 75 mg/kg/ day or DFX in a dose; 20-25mg/kg/d in 2:1 ratio respectively, while those on DFP in START had continued on either DFP or DFX in 1:2 ratio with same doses. Patients were kept on regular transfusion to keep pre-transfusion Hb >8gm/dl. The primary endpoint was safety and secondary endpoints were changes in serum ferritin (SF) and growth enhancement (height, weight) both were assessed quarterly. An informed consent was signed by parents of all patients before start of the study. Statistical significance between DFP and DFX treated groups was calculated via t-test for continuous variables and Fisher's exact test for discrete variables. Results: In the current study; 66% of the children were males, at enrolment; their age ranged from 20-68 (median 38) months; those who were on DFP or placebo in START had received comparable transfusion units (median 11 and 9 respectively) and SF (880 and 1150 ng/ml respectively). At 12 month of continuation on iron chelation 22% and 60% of children on DFP vs 12% and 48% of those on DFX had SF<500 ng/ml or 500- <1000 ng/ml, respectively (p value <0.05);. TDB-T initiated and continued on DFP had the best growth velocity; all had annual growth velocity >5cm (p value <0.01), as well as a significant lower final SF (P value < 0.01). Adverse events were mild and uncommon in both groups. There were no episodes of arthralgia or agranulocytosis in either group. Elevated serum creatinine > 33% from baseline on 2 successive visits were observed in 0% in DFP vs. 12% in DFX. No unexpected, serious, or severe AEs were reported in both groups. Conclusion: Children with TDB-T whether on DFP or DFX showed uncommon mild AEs, with no serious or severe AEs. Patients on DFP after an early start of DFP showed adequate growth velocity and better control of iron-overload on serial measurement of SF; compared with those who were on placebo.
Hamdy: ApoPharma: Honoraria; Amgen: Honoraria; Bayer: Honoraria; Novartis: Honoraria; NovoNordisk: Honoraria; Roche: Honoraria; Takeda: Honoraria.
Author notes
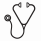 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract